What is a car catalyst: An automobile catalyst or catalytic converter is an exhaust gas emission control device.
A redox catalyst (redox reaction) converts toxic gases and pollutants present in the
exhaust gases of the internal combustion engine into less toxic pollutants.
Catalytic converters are commonly used in internal combustion engines that run on gasoline or diesel.
What is a car catalyst and what are the benefits of using it?
Although catalytic converters are most commonly used in automobile exhaust systems,
they are also used in electric generators, forklifts, mining equipment, trucks, buses, locomotives,
motorcycles, and ships. It is even used in some wood-burning stoves to control the emission of toxic gases.
History of the use of auto catalysts This was the first large-scale introduction of catalytic converters into the American automobile market.
To comply with emissions regulations issued by the US Environmental Protection Agency, most gasoline-powered
vehicles that have entered the market since 1975 are equipped with catalytic converters.
What is a car catalyst and why is important?
These suits are created "in a way" with accoutrements of monocarbonate (CO),
cyclic hydrocarbons (CnHn), carbon dioxide (CO2), and water (H2O) being produced. In 1981, Catalizori Suits
was created by "Ha-H-H" Suits, which was completed by Nitro Accessories.
In Iran, catalysts for Peugeot cars, catalysts for 206 cars,
and catalysts for Teyba are available in different types and at different prices, for example,
the price of a Rio exhaust catalyst varies according to the type of catalyst.
However, 2-way converters are still used in lean engines because 3-way converters require powerful or
stoichiometric combustion to successfully reduce NOx.
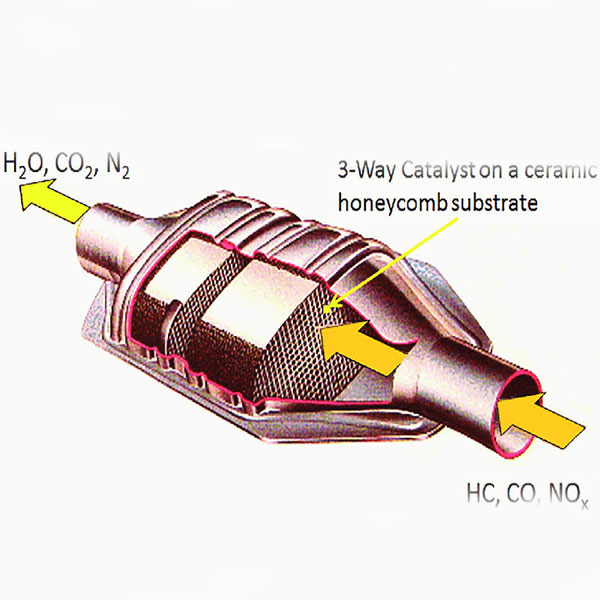
What is a car catalyst ? Car catalyst structure!!
 The catalytic converter is made of various materials.
The catalyst core or bed varies depending on the vehicle. For example, when these devices are used in cars,
The catalytic converter is made of various materials.
The catalyst core or bed varies depending on the vehicle. For example, when these devices are used in cars,
Its core is usually a ceramic block with a honeycomb structure.
Ceramic cores can be cheap if mass-produced. In some applications, integral foils made of a mixture of iron
, chromium and aluminum are used.
Metal cores are less expensive when manufactured for use on smaller tracks, such as sports cars,
where low pressure and reliability under high static loads is essential.
Both of these materials are designed to provide a large surface area to support a catalyzed floor coating.
What is a car catalyst ? car catalyst components
Triggers consist of the following three main parts:
Catalyst: This part consists of metals such as palladium, radium, platinum, gold,
cadmium or a combination thereof.
Naturally, the use of these precious metals increases the price of the exhaust catalyst,
which indicates the importance of maintaining it for a longer life.
Catalyst core: This part is a monolithic ceramic substrate that most often has a honeycomb structure and
is placed in the center of the catalyst.
Oxide layer: This layer consists of aluminum oxide, silica, alumina or titanium dioxide.
The duty of this part is to protect and maintain the catalyst.
catalyst form
That being said, each car catalyst, also known as a catalytic converter,
has three parts: the core, the oxide layer, and the catalyst.
The shape and phase of the catalysts in a chemical reaction can be in phase with the reactants
or in phase with the reactants. Accordingly, stimuli are divided into homogeneous and heterogeneous categories.
Homogeneous catalysts are in co-phase with the reactants.
But its heterocyclic type goes through a different phase with the reactive materials.
The reaction is heterogeneously carried out on their surface. ( تعمیر کاتالیزور )
مرکز تخصصی تعمیرکاتالیزور


ثبت ديدگاه